Why are Stearates added to most supplements?
And why do we recommend that you avoid them completely?
-
Magnesium Stearate is used in over 95% of commercial supplements to speed up manufacturing and improve profit margins.
-
High-speed encapsulation machines typically require the addition of Stearates as a flow agent so massive quantities of mass-market capsules can be manufactured cheaply to ensure higher corporate profits.
-
Tablets typically use a much higher content of Stearates to bind the tablet together.
-
There is no nutritional purpose for adding Stearates to the diet...
-
We do not use Stearates in any of our Functional Formulations™.
-
It takes more time, care and attention to detail to make remedies without magnesium stearate, stearic acid, lubricants, flow agents, fillers, binders, coatings, phthalates, dyes, or other non-nutritive additives, but we believe you are worth it, and the results prove it every day. For clean, concentrated, and comprehensive botanical/nutritional remedies, visit RemedyMatch.com.
What's wrong with eating Stearates?
-
Reduced Bioavailability of Active Ingredients:
Magnesium Stearate slows the digestion of capsules by 65%, taking about 3 times as long to release the contents, delivering the active ingredients further down the digestive tract, reducing or missing key opportunities for absorption. In blending the product ingredients, nutrients and botanicals can also get coated by the Magnesium Stearate, further slowing digestion and reducing bioavailability… Most nutrient absorption occurs in specific portions of the small intestine...
Source:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5736124/
Most tablets use much more Magnesium Stearate than capsules to make them stick together, making them even slower to digest, sometimes even appearing intact in the stool…
-
Reduced Absorption:
Magnesium Stearate forms a sticky biofilm coating the gut mucosal lining, reducing the absorption of nutrients in the diet as well as in supplements.
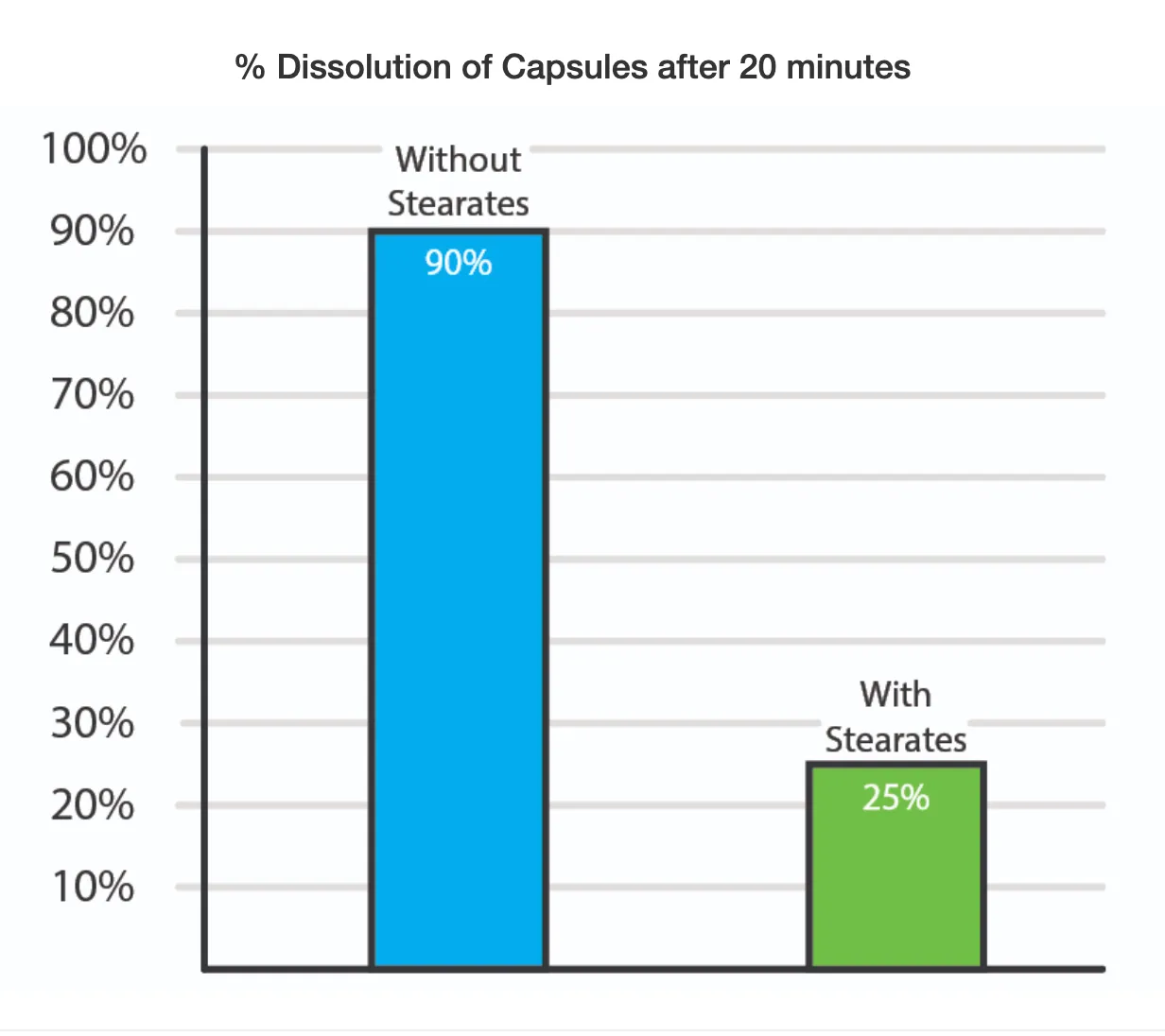
A study published in Pharmaceutical Technology in 1985 showed that capsules dissolve 3.6 times better without Stearates, as shown in the graph below.
-
Gut Irritation:
Magnesium Stearate can irritate the mucosa, increase leaky gut issues and even trigger chronic diarrhea…
All these issues are further compounded if there are any underlying gut issues already, including SIBO, Crohns, Colitis, Diarrhea, Leaky Gut Syndrome, or other common challenges…
The pathophysiology of most common health issues in other body systems begins with undetected issues in the gut.
-
Cellular Integrity:
Stearic Acid, the anion released when Magnesium Stearate is digested, "may enhance membrane rigidity to an extent that plasma membrane integrity is significantly impaired, leading to a loss of membrane potential and ultimately cell function and viability."
Source:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1384169/
-
Immunity:
Magnesium Stearate is immunosuppressive by suppressing the activity of the T-cells. “…stearic acid (18:0) selectively inhibits T-dependent immune responses…"
Source:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1384169/
-
Impurities:
Magnesium Stearate is made with toxic oils such as hydrogenated cottonseed oil and palm oil. These sources are frequently genetically modified (GMO). The source oils can have the highest pesticide residue levels of all commercial oils due to the heavy spraying of cotton crops. Processing involves high heat, high pressure, and metal catalysts, adding more potential for detrimental effects and contamination...
-
Hazardous Substance:
In addition to its use in making most commercial supplements, Magnesium Stearate is used in manufacturing ammunition and as a paint and varnish drier...
The Environmental Protection Agency (EPA) requires filing a Material Data Sheet for the manufacture and transport of Magnesium Stearate, which is classified as a “Hazardous Substance.”
© Copyclaim 2023
Remedy Match LLC, DBA Healing Oasis
[email protected]
PO Box 126 Hilo, Hawai'i-Kingdom [96721]
+1 (808) 217-9647
[*"The statements herein have not been evaluated by the Food and Drug Administration. This is not intended to diagnose, treat, cure, or prevent any disease."] T.D.C.
